Blog
What Are All the Types of Chemicals Explained?
Chemicals play a vital role in various industries today. Different types of chemicals serve specific functions, from pharmaceuticals to agricultural products. The "all chem" sector is expansive, encompassing numerous classifications and uses. According to the American Chemical Society, the chemical industry contributed over $750 billion to the U.S. economy in 2022 alone. This significant impact underlines the importance of understanding chemical classifications and their applications.
Dr. Emily Johnson, a leading expert in the "all chem" field, once stated, “Diversity in chemicals is essential for innovation and progress.” This emphasis on diversity highlights the need for a comprehensive view of available chemicals. Knowing their unique characteristics can lead to better product development and safety measures.
However, this complexity also brings challenges. Misunderstanding chemical types can lead to safety hazards. The industry must prioritize education on chemical classifications. Encouraging deeper awareness will help mitigate risks. The intricate landscape of chemicals demands our attention and careful consideration to harness their full potential.
Types of Chemicals: An Overview of Chemical Classification
Chemicals are everywhere, making up our world in various forms. Classifying them is essential for understanding their properties and uses. Broadly, we can categorize chemicals into several types: organic, inorganic, physical, and biochemicals. A report by the International Chemical Industry Association states that organic chemicals account for roughly 75% of all chemical production globally.
When we talk about organic chemicals, we refer to compounds primarily made of carbon. These often form the basis for fertilizers, plastics, and pharmaceuticals. In contrast, inorganic chemicals primarily encompass metals and minerals. They are vital in construction, electronics, and more. A report from the American Chemical Society highlights that inorganic chemicals represent about 25% of the global chemical market, reflecting their essential role in various industries.
Despite these classifications, overlaps and gray areas exist. For instance, biochemicals, though often grouped with organics, present unique properties. They arise from living organisms and play crucial roles in biotechnology. Yet, challenges remain in classification, especially with emerging compounds. The constantly evolving landscape raises questions about how we categorize these substances effectively. It's an area ripe for discussion and reflection in the scientific community.
What Are All the Types of Chemicals Explained? - Types of Chemicals: An Overview of Chemical Classification
| Chemical Type | Description | Examples |
|---|---|---|
| Organic Compounds | Compounds primarily made of carbon and hydrogen, often containing functional groups. | Methane, Ethanol, Acetic Acid |
| Inorganic Compounds | Compounds that do not contain carbon-hydrogen bonds; typically found in minerals. | Sodium Chloride, Water, Ammonia |
| Acids | Substances that donate protons (H+) in solution; have a sour taste. | Hydrochloric Acid, Sulfuric Acid |
| Bases | Substances that accept protons or donate electron pairs; have a bitter taste. | Sodium Hydroxide, Ammonium Hydroxide |
| Salts | Ionic compounds formed from the reaction of an acid and a base. | Sodium Chloride, Potassium Nitrate |
| Gases | Substances that exist in gaseous state at room temperature; low density. | Oxygen, Carbon Dioxide, Nitrogen |
| Liquids | Substances that are in liquid state at room temperature; have definite volume. | Water, Ethanol, Acetone |
| Solids | Substances that maintain a fixed shape and volume; particles are closely packed together. | Iron, Ice, Table Salt |
Organic Chemicals: Characteristics and Examples
Organic chemicals are everywhere. They form the basis of life. Characteristics of organic chemicals include carbon-containing compounds. These compounds often include hydrogen, oxygen, and nitrogen. Organic chemicals can be simple, like methane, or complex, like proteins.
Examples of organic chemicals range widely. Alcohols, such as ethanol, are commonly used. Carbohydrates like glucose provide energy. Amino acids are building blocks for proteins. Each example showcases unique properties and functions. They play crucial roles in biology and industry.
Tip: When working with organic chemicals, always ensure proper labeling and storage. This minimizes risks and prevents accidents. Understanding the properties of each chemical is vital for safety.
Some organic compounds can be hazardous. They may react unpredictably in certain conditions. Awareness of their properties can prevent mishaps. It's essential to approach the use of organic chemicals with caution. Awareness leads to smarter decisions.
Inorganic Chemicals: Fundamental Properties and Categories

Inorganic chemicals play a crucial role in our daily lives and industrial processes. They are not based on carbon, unlike organic chemicals. This category includes metals, salts, and minerals. According to the International Journal of Environmental Research, inorganic compounds comprise about 90% of all known chemical substances. This highlights their significance in various sectors, including agriculture and manufacturing.
These compounds can be divided into several categories. Metals such as iron and copper are essential in construction and electronics. Salts, like sodium chloride, are vital in food preservation and chemical reactions. A report from the U.S. Geological Survey estimates that 90 million tons of inorganic fertilizers are used globally each year. These fertilizers boost agricultural productivity, yet their excessive use can harm soil health.
Understanding the behavior of inorganic chemicals is essential. They have distinct properties, such as high melting points and electrical conductivity. However, the environmental impact of their production and disposal requires serious reflection. Striking a balance between their utility and sustainability is a challenge. Effective regulations and practices are needed to minimize negative effects while reaping their benefits.
Biochemicals: Role in Biological Processes and Applications
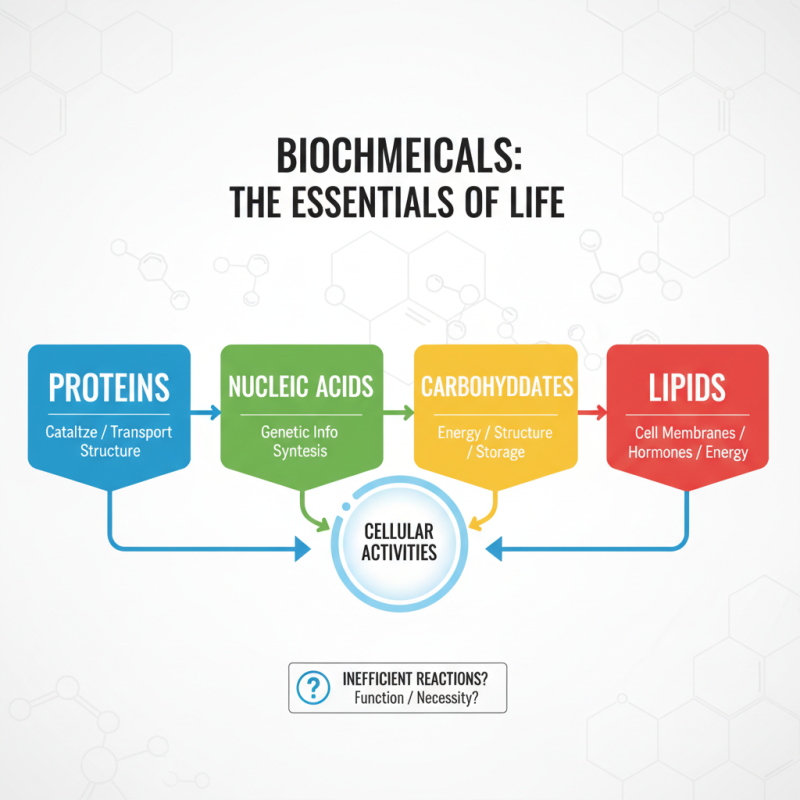
Biochemicals play a crucial role in biological processes. They are organic molecules essential for life. Proteins, nucleic acids, carbohydrates, and lipids fall into this category. Each type has a unique function that supports cellular activities. For instance, proteins catalyze reactions and transport molecules. Some of these reactions are not efficient, raising questions about their necessity.
Enzymes, a type of protein, act as catalysts. They speed up reactions but may not always be effective. Some enzymes require specific conditions to work properly. Without the right temperature or pH, they can be inefficient. This can lead to cellular stress or malfunction.
Nucleic acids, like DNA and RNA, store genetic information. They guide the synthesis of proteins. However, mistakes often occur during replication. Such errors can lead to mutations and diseases. This highlights the fragility of life at a molecular level. Overall, biochemicals are vital, yet their complexities and limitations provoke reflection on our understanding of biology.
Specialty Chemicals: Uses and Industries Involved
Specialty chemicals play a crucial role in various industries. These chemicals are tailored for specific applications. They are distinct from commodity chemicals, which are produced in bulk. According to market research, the global specialty chemicals market was valued at approximately $1 trillion in 2023. This sector is projected to grow at a compound annual growth rate of around 4.5% over the next five years.
The uses of specialty chemicals span numerous industries. For instance, they are vital in agriculture, used in fertilizers, pesticides, and herbicides. In the automotive sector, specialty coatings enhance vehicle durability and aesthetics. Additionally, specialty chemicals are instrumental in personal care products, offering enhanced performance and functionality. The formulation of these products often requires precise control over chemical properties, which may lead to unexpected challenges. In a recent study, almost 30% of respondents reported issues related to formulation consistency and ingredient sourcing.
Despite advancements, the specialty chemicals industry faces hurdles. Supply chain disruptions can impact production schedules. Fluctuations in raw material prices also pose risks for manufacturers. Adapting to these market dynamics is essential. Industry professionals must stay informed about innovations and regulatory changes. As the sector grows, there is a continued need for sustainable practices and responsible sourcing.
Specialty Chemicals: Uses and Industries Involved
Related Posts
-

How to Choose the Right Chemical Raw Materials for Your Projects
-

How to Choose the Right Chemical Producers for Your Business Needs?
-

Top 10 Specialty Chemicals Driving Innovations in Various Industries
-

Top 10 Specialty Chemicals Transforming Industries Today?
-

Understanding Solvent Pigments in 2025 Top Applications and Innovations
-

2026 Best Organic Pigments for Art and Design Applications?

